Thermo Example 1 © 2000 D.P. Maloney
One hundred and fifty joules of heat are added to 75 g of a material whose specific heat is 1.25 J/g Co. If the material’s initial temperature was 43oC, what is its final temperature?
Given:
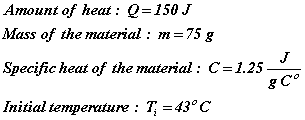
Unknown: ![]()
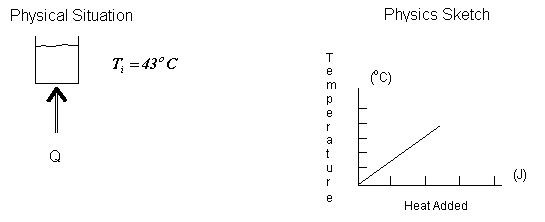
Physical Principles and/or Ideas: Temperature change of a material when heated, definition of specific heat.
Solution: This problem is reasonably straightforward. A material is being heated without undergoing any change of state. In such a situation, all of the heat added to the material goes into increasing the temperature of the material. The rate at which the temperature increases and the actual temperature change are determined by the mass of the material and the material’s specific heat. Using this relation, we find:
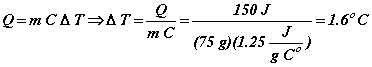
So,
![]()
 Physics Department [Return to Examples]
Physics Department [Return to Examples]